
 |
 |
|||
| An introduction to growing and differentiating Trypanosoma brucei in vitro | |||||
| I have assembled quite detailed information about the history and current methods of growing Trypanosoma brucei bloodstream and procyclic forms in liquid media and on agarose plates. I hope you find at least some elements of this effort to be interesting and useful. | |||||
| I have written A personal commentary on the history and current art of culturing Trypanosoma brucei available in browser-viewable html and pdf formats. A few key publications mentioned in this commentary, which are not otherwise available online, have been scanned with OCR software and converted into pdf files, as a service to those who might find them useful. These are mostly articles that I authored, to minimize copyright issues. The files may contain OCR errors. Text conversion was used, rather than scanned images, to minimize file sizes.
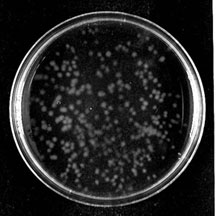
I have prepared a chart comparing the (quite different) compositions of commonly used culture media and protocols for preparing media fior culturing bloodstream (BF) and procyclic (PF) forms and for differentiating from BF to PF. A detailed protocol for agarose plating/cloning of BF trypanosomes pdf was written by Vern Carruthers and slightly modified by Kevin Tan. This protocol has now been supplemented with a Quicktime™ movie (click to open in a new window) showing how to spread plates and illustrations (below) of how to photograph your colonies, all prepared by Kevin Tan. You may need to download Apple's free Quicktime plugin to view the video. In 2004, McCulloch, Vassella, Burton, Boshart and Barry published a comprehensive review entitled Transformation of Monomorphic and Pleomorphic Trypanosoma brucei as a chapter in "Genetic recombination: reviews and protocols". The chapter can be purchased from the publisher, Humana Press (a division of Springer). As with any book chapter, it does not contain the most up-to-date information on some aspects, especially in regard to efficiently cloning Procyclic Forms, where we are indebted to members of the Paul Englund laboratory for a protocol (below) that allows procyclics to be cloned with about 50% efficiency in the absence of any requirement for conditioning of the medium. Additional Advice on Cloning Procyclic Forms Procyclic forms initially proved more difficult to clone than bloodstream forms. They originally appeared not to grow at low concentrations (below ~104/ml, and several labs, including ours, adopted the use of 'conditioned medium' to improve cloning efficiency. Some labs even added live wild-type trypanosomes to the intial culture, when cloning transfected ones. Liz Wirtz provided the original advice that has been retained at the end of this section, as an addition to the description in the Methods section of Wirtz et al 1999. In 2004, however, we adopted, with great success, a simpler and robust protocol provided by Shawn Motyka and Paul Englund. The protocol was included in a July 2002 publication (Klingbeil, M. M., S. A. Motyka, and P. T. Englund. 2002. Multiple mitochondrial DNA polymerases in Trypanosoma brucei. Mol Cell 10:175-186 DOI PMID), which says PF were “…cultured in SDM-79 medium containing 15% fetal bovine serum ... cloned by limiting dilution in … 96-well tissue culture plates at 1 cell/ml under 5% CO2 at 27°C (cloning method from W. Gibson, University of Bristol) … plating efficiencies ranging from 30%–60%”. The key element, giving what they described (personal communication) as a "drastic improvement in cloning efficency", was raising the serum level from 10% to 15%. This protocol gave >50% cloning efficiency in our hands, more than twice as good as we obtained with conditioned medium in 10% serum. The cells are diluted to 5/ml in SDM containing bicarbonate (as it should) and 15% serum, and 100 microliter aliquots are distributed into 96-well flat-bottomed plates. The plates are incubated unsealed in a properly humidified CO2 incubator (proper incubator setup is highly technical: reading the manual is essential). Clouding of the wells (about 20 per plate), indicating trypanosome growth, is usually seen after 12 days, at which time the samples are diluted to a volume of 1 ml in medium with 10% serum. Over a two-week period, Shawn reports about a 20% evaporation from wells around the perimeter of the plate, but this does not appear to be a problem (my original HX25 procyclic defined medium was very hypertonic, as the tsetse midgut may be). Liz Wirtz's original advice for cloning procyclic forms, for anyone who might still find it interesting or useful is as follows. To prepare conditioned medium, cells from a late log-phase culture (7 to 9 x 10^6/ml) in SDM79 were centrifuged and the supernatant filtered. The filtered conditioned medium can be stored a few weeks at 4°C or frozen. Appropriate combinations of inhibitors and/or inducers (hygromycin, G418, phleomycin +/-Tc) must be added to the conditioned medium before use. 1 ml of undiluted culture is placed into the first row of wells and then a 1:1 dilution (0.5 ml original culture plus 0.5 ml conditioned SDM-79) in the 2nd row. The 3rd and 4th rows contained serial dilutions of the second row (1:1 or 1:2), in conditioned SDM79. Phleomycin eliminates untransformed cells in about 7 days. By about 6 days there is a pronounced difference in numbers of viable cells between transfected and negative controls. pLew100 can be used as a positive control. Positive wells must be allowed to become very dense before feeding. The medium will look very yellow and ugly, but it is imperative to resist the urge to feed them. Dilution can kill more transfectants than the added inhibitors! Only when wells are literally teeming with cells (>4 x 10^6/ml : about day 9 or day 10 for pLew100 transformants) should they be fed. The volume in the well must be increased by no more than 1:1 dilutions. When the volume reaches 2 ml, cells can be transferred to a flask and be progressively but not aggressively diluted. If nascent procyclic clones are diluted below about 3 x 10^6/ml, their growth will slow or they will die. Selected historical publications are being made available as pdf files on this site. Cultivation of Trypanosoma brucei sspp. in semi-defined and defined media (1973) Cross, GAM & Manning, JC. Parasitology 67:315-31 (pdf 488k). |
|
 |
 |